
( B) Schematic presentation of the olfactory system of H. The PNs in other antennal-lobe tracts (ALTs) are not shown in this panel. After innervating the Ca, the medial-tract PNs project into distinct sub-regions in the lateral protocerebrum according to the behavioral significance of the relevant odor signal. Instead of encoding the identity of stimuli, the Kenyon cells process signals of positive versus negative valence into different terminal clusters ( Aso et al., 2014a Aso et al., 2014b). The neuron connectivity in Ca has a semi-random topographical distribution, where signals are segregated according to the nature of sensory input, for example, pheromone and general odor PNs have their terminals located at the inner and outer part of calyces, respectively ( Zhao et al., 2014), but connections between the projection neurons and the Kenyon cells are random ( Caron et al., 2013). The medial-tract PNs first project to the calyces of the mushroom body (Ca), where they contact Kenyon cells. Neurons tuned to food odors versus pheromones are portrayed in green and red, respectively. AL projection neurons (PNs) passing along the most prominent tract, the medial AL tract (mALT) have dendrites in a single glomerulus and receive direct input from one OSN class. ( A) All olfactory sensory neurons (OSNs) that express the same odorant receptor project their axons to the same antennal-lobe (AL) glomerulus. Thus, at the level of the lateral protocerebrum, it appears that odor cues are represented in different widespread sub-domains that display a form of spatial pattern according to behavioral significance, including valence. 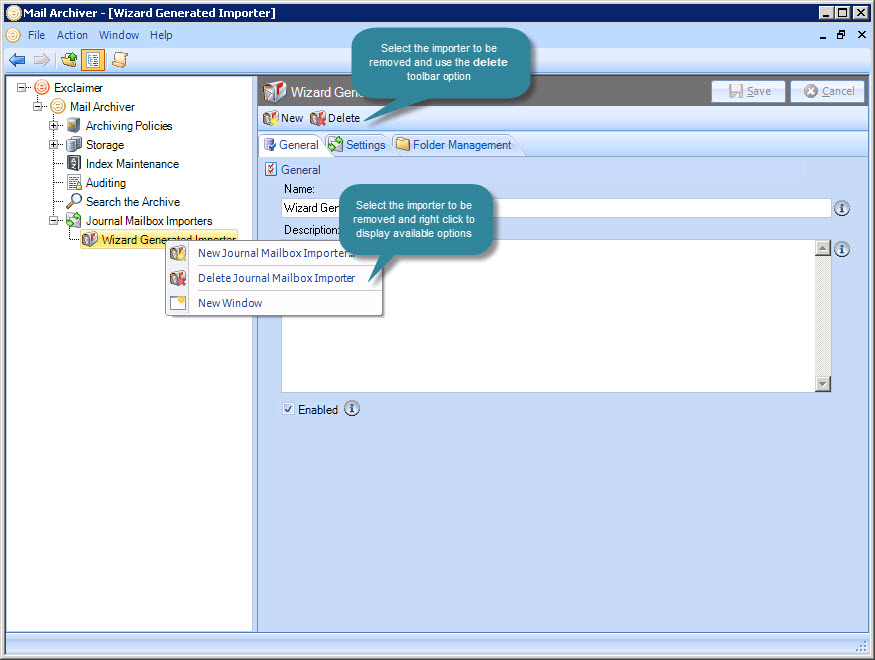
In the fruit fly, such spatial separation is shown to include even attractive versus repulsive odor signals ( Grabe and Sachse, 2018 Min et al., 2013). Signals inducing different behaviors, such as pheromones versus food odors, are segregated in the lateral protocerebrum both in fruit fly and moths ( Grabe and Sachse, 2018 Homberg et al., 1988 Zhao et al., 2014). In contrast to the semi-random neuronal connectivity in another higher-order olfactory center of the insect, the mushroom body calyx (mammalian piriform cortex analog Su et al., 2009), the neuronal wiring in the lateral protocerebrum is characterized by a form of spatial clustering relying on behavioral significance (for details, see Figure 1A). This brain region shares many neural principles with the mammalian cortical amygdala ( Miyamichi et al., 2011 Sosulski et al., 2011). In moth, pheromone-evoked behaviors are linked to a hardwired circuit in the lateral protocerebrum, including the lateral horn ( Insect Brain Name Working Group et al., 2014 Martin et al., 2011). 
With a relatively simple brain and a restricted number of associated odors evoking opposite innate behaviors, that is, attraction and aversion, the insect pheromone pathway is an optimal system to address this question. Understanding how these chemosensory circuits translate signals with different hedonic valences into behavior is an essential issue in neuroscience. Olfactory circuits serve a central role in encoding and modulating sensory input from the natural surroundings. Our data suggest that the neurons’ widespread projections are organized according to behavioral significance, including a spatial separation of signals representing attraction versus inhibition – however, with a unique capacity of switching behavioral consequence based on the amount of the minor components. 
In this study, we traced physiologically identified medial-tract projection neurons from each of the three male-specific glomeruli with the aim of mapping their terminal branches in the lateral protocerebrum. From the antennal-lobe glomeruli, the information is conveyed to higher olfactory centers, including the lateral protocerebrum, via three main paths – of which the medial tract is the most prominent. In Helicoverpa armigera, three male-specific glomeruli receive input about three female-produced signals, the primary pheromone component, serving as an attractant, and two minor constituents, serving a dual function, that is, attraction versus inhibition of attraction. The pheromone system of heliothine moths is an optimal model for studying principles underlying higher-order olfactory processing.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
